LoFV and collaborators release new bat cell lines to the research community
The first bat cell line was deposited with ATCC in 1967.
Anybody studying bat viruses and immunology knows there are not enough publicly available resources. You can only buy 3 bat cell lines at the moment and they aren’t suitable for many viruses. This lack of resources has become such a problem for researchers studying innate immune responses to infection and bat tolerance of pathogenic viruses that the NIH has made several calls for the scientific community to develop more laboratory reagents for bat research.
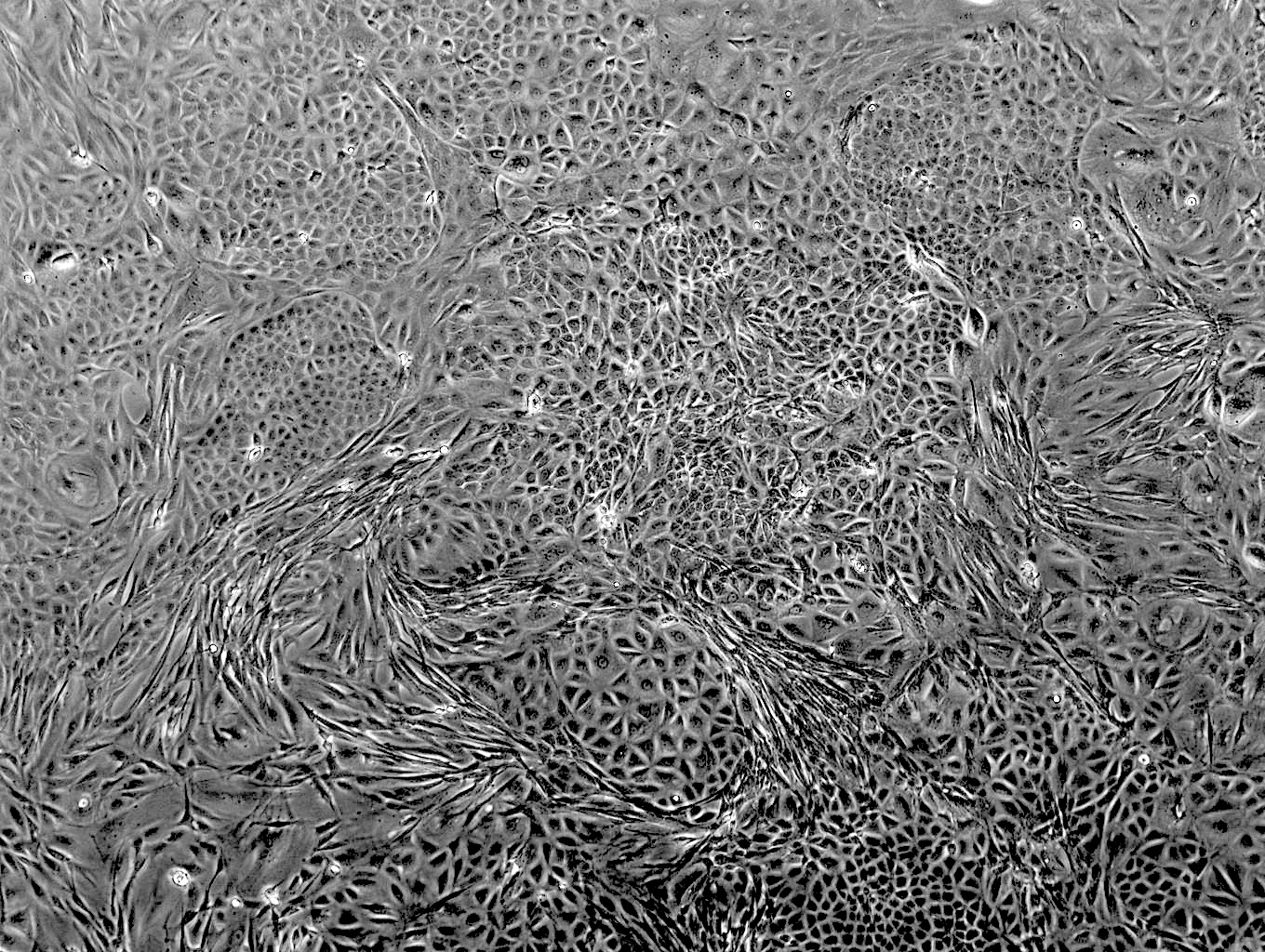
As part of a 3-year collaboration with Stephanie Seifert at WSU, Arinjay Banerjee at VIDO, Rohit Jangra at LSU, and Alison Kell at UNM, we developed new cell lines from Carollia bat tissues specifically with the goal of making new tools for the research community. The resulting clonal, immortalized cell lines grow under standard lab conditions without the need for fancy medias, or expensive culturing practices. They can be infected with a wide range of viruses, including MERS-CoV, orthohantaviruses and VSV and respond to interferon stimulants.The best part of all is that we made our top performing bat cell lines from this study available through BEI resources/ATCC! We encourage you to use these reagents for your next molecular bat study!
Take a look at our new preprint and don’t hesitate to contact us for more information or to request other reagents from this study that are not available through BEI. The hold for these reagents has now been lifted so they will be available soon.
Special thanks to Christine Portfors at the WSU campus in Vancouver, WA for providing bat tissues and to all the talented team members who helped us develop these new tools.
We would also like to thank Dr. Jay Hooper at USAMRIID for generously providing orthohantavirus M segment genes and B. Haagmans and R. Fouchier, Erasmus Medical Center, for providing MERS-CoV (isolate hCoV-EMC/2012). Research reported in this publication was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health (NIAID/NIH) under Award Numbers 1R21AI169527 (M.L. and A.B.), R 21AI156482 and P20GM134974 (R.K.J.), and Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grant (RGPIN-2022-03010) awarded to A.B. This work was partly supported by an NSF Biology Integration Institute grant under award numbers NSF DBI 2021909 and 2213854 (S.N.S). V.G. is supported by an NSERC scholarship (#569587-2022). VIDO receives operational funding from the Government of Saskatchewan through Innovation Saskatchewan and the Ministry of Agriculture, and from the Canada Foundation for Innovation through the Major Science Initiatives Fund.