Congrats Chad! His article (#11!) in Nat. Chem. is highlighted in C&EN magazine!!! Check out the first Pr5+ molecular compound in a singlet ground state with significant multiconfigurational character: https://cen.acs.org/materials/inorganic-chemistry/Praseodymium-powers-435/103/web/2025/04
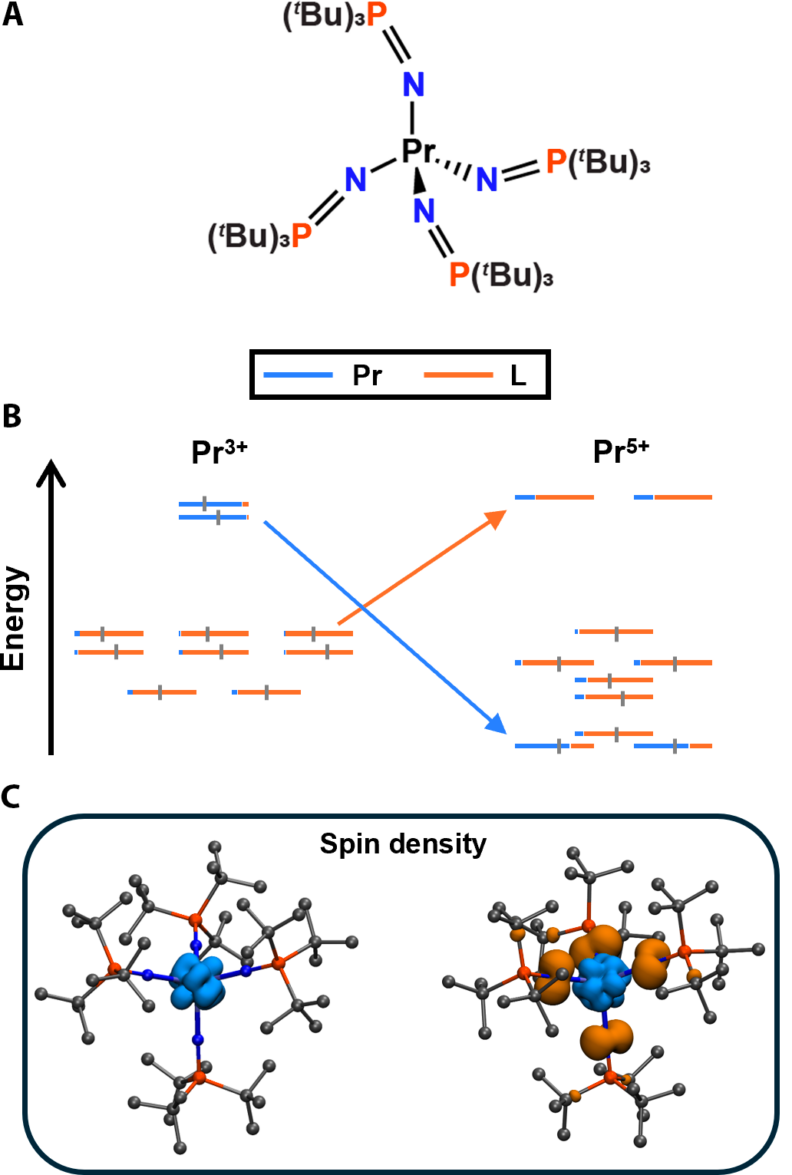
Our calculations reveal the presence of an inverted ligand field—an unusual electronic configuration where the lowest unoccupied molecular orbitals are predominantly ligand-based rather than metal-centered. This finding challenges conventional oxidation state descriptions and links high-valent lanthanide chemistry to transition-metal systems with similar electronic behavior.
Computational results indicate a singlet ground state with significant multiconfigurational character in [Pr5+(NPtBu₃)₄]+ (tBu = C(CH3)3), where occupied metal-dominant orbitals (avg. %Pr = 69.9) reside at lower energies than ligand-dominant ones. Unlike the classic ligand field observed in the previously reported Pr3+ and Pr4+ imidophosphorane complexes, the antibonding orbitals in Pr5+ exhibit greater ligand contributions (avg. %ligand = 74.5). Additionally, a notable spin density on the Pr center is counterbalanced by the four nitrogen atoms, further supporting the unique electronic structure and redox properties of early lanthanides (see Figure).
By combining experimental results with advanced computational modeling, this study underscores the vital role of theoretical chemistry in unraveling the complex electronic structures of f-block compounds and establishes a key connection in understanding the bonding of high-valent metal complexes across the periodic table.
