Material Applications
Organic Solar Cells for Renewable Energy, Medicine, and Agriculture

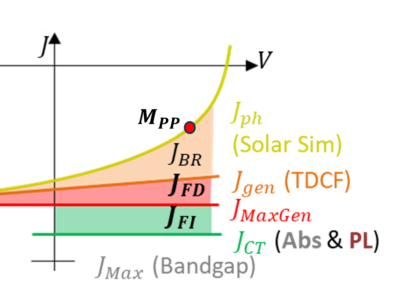
Solar cell technologies are one of the most promising energy solutions of the 21st century offering the promise of renewable energy to fill the needs of our ever growing society. A majority of solar cell solutions are costly to mass produce and create toxic waste products dampening their usefulness in fighting the current climate crisis. Organic Solar Cells (OSCs) manufactured from organic molecules and polymers are cheep, clean and easy to produce making them a major disruptive technology to this billion dollar industry. Unlike traditional solar cells, organic solar cells are manufactured from inexpensive and abundant and inexpensive materials that are easy to manufacture and upscale. A prime example of this is roll to roll printing, where ink-jet printers are repurposed to print solar cell material. Another key example is spray coating, where the solar cell materials are literally spray painted onto electrodes. The nature of the organic solar cell materials not only allows for these interesting process methods, but also provides a degree of tune-ability to the kinetic and optical properties of these materials. Allowing for bendable and transparent solar cells with interesting applications in agriculture.
The efficiency of OSCs in converting sun light photons into free charges has increased drastically since their discovery leading to near parity with traditional counterparts. This increase is driven by the understanding of charge dynamics though hidden nano-scale structures in these materials. Our group specializes in understanding the complete dynamics excited charges take as they navigate the torturous 3d landscape in a material to make it to the electrodes and generate power. This requires a multitude of in house experiments and analysis methods to quantify where charges are lost along the journey. We pair this analysis with National Lab Synchrotron techniques to measure OSC nano-structures allowing us to get a full picture of this 3d landscape. This knowledge is then used by others in the field to design new molecules, improve solvent processing methods, and make other changes that drive the performance of these devices forward.
Perhaps the larges jump in device performance has been the emergence of the Ternary OSCs (TOSCs) created by the mixing of three material components. These solar cells out compete all others but no one yet knows what fundamental physics processes lead to the the emergence of their high efficiency. Due to the presence of the guest molecule, TOSC nano-structures is much more complex making it infeasible to optimize them using typical trial-and-errors methods. This is why our groups current primary goal in the OSCs space is to understand the dynamics and nano structures in responsible for the high performance of TOSC devices. We are currently developing our capabilities for novel in house experimental methods, along with X-ray based nano probes to unravel this mystery. By combining these methods, we will be able to predict the nano-structures responsible for the high-efficiency of TOSC devices allowing this technology to revolutionize renewable energy and other important applications.
Thermodynamics of Mixing
Most of our organic photovoltaic (OPV) devices are fabricated by dissolving conductive molecules in organic solvents such as isopropyl alcohol, acetone, or even chloroform. These molecules are combined in carefully optimized ratios to maximize device performance. The resulting solution is then coated onto a substrate, where the solvent rapidly evaporates, leaving behind a thin film roughly 100 nm thick. During the brief 30-second deposition process, molecular interactions with both the solvent and each other drive the formation of mixed domains. These domains provide efficient pathways for electrons and holes to hop between molecules and ultimately reach the electrodes to be harvested for electricity!
Right now, one exciting direction in our lab is studying ternary OPVs, which use three different molecules instead of just two, and these systems are setting new records for solar cell performance. To understand why, we need to answer the same big question: how do these molecules actually like to mix with one another? One powerful tool we use is a Scanning Transmission X-ray Microscope, which lets us zoom in and measure the composition of our thin films. Since each molecule has its own unique “fingerprint” in the way it absorbs X-rays, we can tell where each one is located in the film and how well they are blending. This gives us a clearer picture of what’s happening on the nanoscale and helps guide us toward even better-performing devices.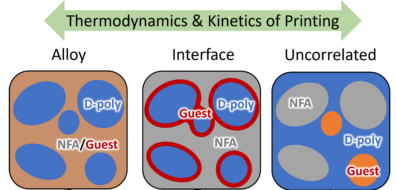
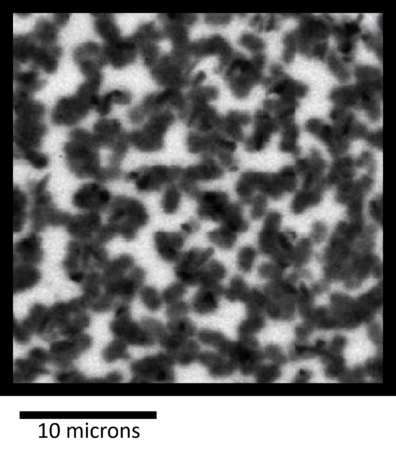
Another way we study how molecules interact is through contact angle measurements. This technique looks at how a droplet of liquid sits on a surface—whether it spreads out like water on glass or beads up like rain on a raincoat. From that simple angle, we can calculate the surface energy of the material. By comparing the surface energies of two different molecular surfaces, we can figure out their free interaction energy—basically, how much they “like” or “dislike” sticking together. It’s a surprisingly straightforward experiment that gives us valuable insight into the compatibility of the materials we use in our solar cells.


